Antifertility Effect of Sperm Agglutinating Factor Isolated from Serratia marcescens: An In vivo Study
Prabha V Negi S, Chauhan A, Vander H, Rana K and Thaper D
Prabha V* Negi S, Chauhan A, Vander H, Rana K and Thaper D
Department of Microbiology, Panjab University, Chandigarh, India
- *Corresponding Author:
- Prabha V
Professor, Department of Microbiology,
Panjab University, Chandigarh-160014,
India
Tel: 91-172-2534140
Fax: 91-172-2541770
E-mail: satishvijay11@yahoo.com
Received Date: April 06, 2018; Accepted Date: August 23, 2018; Published Date: August 30, 2018
Citation: Prabha V, Negi S, Chauhan A, Vander H, Rana K, et al. (2018) Antifertility Effect of Sperm Agglutinating Factor Isolated from Serratia marcescens: An In vivo Study. J Reproductive Endocrinol & Infert. Vol.3 No.1:5
Abstract
The standard strain of Serratia marcescens causing sperm agglutination in vitro leading to infertility in female mice without causing inflammation was already available in the laboratory. So, to investigate the underlying mechanism and its role in infertility, the corresponding sperm agglutination factor was isolated. As only the washed cells, and not the supernatant, showed sperm agglutination so the washed cells were sonicated and the proteins in the supernatant were precipitated out with ammonium sulphate at the saturation of 60-80% and purified by gel permeation chromatography followed by ion exchange chromatography. The sperm agglutination factor (SAF) was found to be a ~54 kDa protein. SAF at a concentration of 60 μg was able to cause 100% agglutination of mouse spermatozoa within 30 min of incubation at 37°C, whereas a concentration of 80 μg resulted in instant loss of viability of mouse spermatozoa and major decrease in Mg++ ATPase activity was observed at a concentration of 100 µg. Further, in vivo studies demonstrated that a single intravaginal administration of SAF (10 μg) before mating completely prevented conception in female mice. Thus, SAF might be one of the reasons for female infertility and with its excellent spermicidal activity, it could be developed as a potent vaginal contraceptive in future.
Keywords
Spermatozoa; Serratia marcescens; Agglutination; Fertility outcome
Introduction
Infertility is the subject of worldwide interest and importance in clinical practice and research which affects both men and women in their reproductive age [1]. It is defined as failure to conceive within one year despite well timed unprotected sexual intercourse [2]. According to clinical data, throughout the world, infertility afflicts a total of 80 million people, not just medically but psychologically too [3]. The factors responsible for female infertility are many and under certain circumstances a combination of factors may be present. These factors can be broadly classified into two categories: non infective and infective causes. Non infective causes include anatomical, hormonal and environmental factors whereas infective causes include colonization of reproductive tract with various microorganisms [4].
Amongst these factors leading to infertility, infections of the genital tract are recognized as one of the major risk factors. It is estimated that these infections account for 30-40% of the infertility cases worldwide [5]. A wide array of microorganisms that have been involved in these infections include sexually transmitted pathogens, such as Chlamydia trachomatis, Neisseria gonorrhoeae, Mycoplasmas, Treponema pallidum, Gardenella vaginalis, Herpes simplex virus, Trichomonas vaginalis or typical uropathogens, such as Escherichia coli, Staphylococcus aureus, Enterococcus faecalis, Pseudomonas aeruginosa, Klebsiella pneumoniae, Enterobater spp. and S. marcescens [6]. The molecular mechanism by which the sexually transmitted microorganisms influence fertility is well established. But barring sexually transmitted microorganisms that are known to cause infertility, the role of other bacteria in infertility is still divisive. Though a number of uropathogens have been demonstrated to elicit detrimental effects on sperm parameters in vitro, but their exact role in female infertility remains elusive.
In an earlier work done in our laboratory, infertility as a consequence of sperm impairing S. aureus and E. coli and their corresponding factors has been observed. Also, standard uropathogenic strain of sperm agglutinating S. marcescens, upon intravaginal administration, rendered the female mice infertile [7]. The underlying mechanism for infertility was conjectured to be the sperm impairing properties of this organism. Hence, the present study was intended with an aim to isolate and purify sperm agglutination factor from S. marcescens and check its effect on fertility outcome in female mice.
Materials and Methods
Microorganisms
The standard strain of Serratia marcescens (MTCC 7641) used in the present study was already available in the laboratory and it was found to be capable of impairing the sperm motility in vitro.
Experimental animals
Sexually mature, 4-5 week old female (22 ± 2 g) BALB/c and 5-6 week old male (25 ± 2 g) mice used in the present study were maintained under standard laboratory conditions in propylene cages bedded with clean rice husk at 20-25°C, in well aerated animal room of the Department of Microbiology, Panjab University, Chandigarh. All the animals were given standard pellet diet and water ad libitum. Animals were allowed to acclimatize to the new housing conditions for at least one week. All the experimental protocols were approved by the Institutional Animals Ethics Committee of the Panjab University, Chandigarh vide letter no PU/ IAEC/S/16/142. The experiments were performed in accordance with the guidelines of the Committee for the Purpose of Control and Supervision of Experiments on Animals (CPCSEA).
Extraction and purification of sperm agglutination factor (SAF)
72 h old culture of Serratia marcescens grown in BHI broth was centrifuged at 10,000 rpm for 15 min at 4°C. The cells were washed twice with sterile PBS and were resuspended in the same buffer. For extraction of SAF, the cell suspension was the subjected to ultrasonication (15 cycles of 30s each with 60s interval in between). Both the cell debris and the sonicated supernatant were checked for spermagglutinating activity. As the spermagglutinating activity was present in the sonicated supernatant, hence ammonium sulphate was used for salting out of the protein. The supernatant so obtained was subjected to ammonium sulphate precipitation so as to achieve 20, 40, 60, 80, and 100% saturation. The precipitates were dissolved in a minimum amount of PBS (50 mM, pH 7.2) and the precipitated protein was dialyzed against PBS under cold conditions and checked for sperm agglutination.
Molecular sieving
For the purification of SAF, molecular sieve chromatography using Sephadex G-200 column (2 cm x 31 cm) equilibrated and eluted with PBS was employed. The precipitated and dialyzed protein dissolved in PBS was loaded on to the column. Fractions of 3 ml each were collected and the absorbance of each fraction was read at 280 nm. Only those fractions which showed the agglutination activity were pooled and concentrated against polyethylene glycol at 4°C.
DEAE-cellulose column chromatography
The pooled and concentrated fractions obtained after molecular sieve chromatography through G-200 were passed through DEAE cellulose and eluted with 0.05, 0.1, 0.2, 0.4, and 0.6M NaCl dissolved in PBS (50 mM, pH 7.2). Fractions of 4 ml each were collected and absorbance was read at 280 nm. The fractions causing agglutination of spermatozoa were pooled and concentrated using PEG at 4°C.
Determination of molecular weight of SAF
The molecular weight of the protein was determined by sodium dodecyl sulphate polyacrylamide gel electrophoresis (SDS-PAGE) using the standard molecular weight marker 21. A 10% gel was prepared and after the gel was run, it was stained using coomassie brilliant blue and post destaining, the molecular weight was estimated.
Impact of SAF on various mouse sperm parameters: in vitro studies
Sperm motility: Sperm motility was determined by the method of Emmens, 1947 [8].
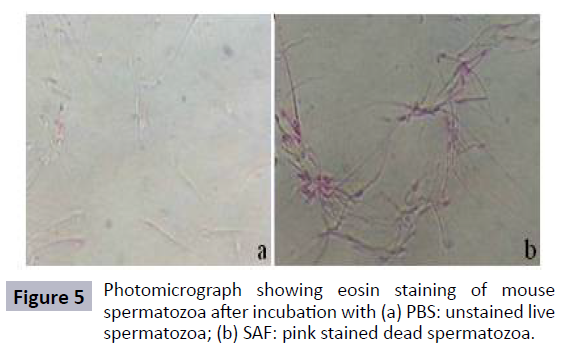
Sperm viability: In order to estimate the percentage of viable spermatozoa, an equal volume of mouse spermatozoa was mixed with 0.5% eosin and examined under the light microscope at 400X magnification.
Sperm Mg++ ATPase activity
Mg++ ATPase activity of spermatozoa was studied according to the method [9,10] Tris-HCl (0.2 M, pH 7.6) washed spermatozoa (1 × 108/mL) were sonicated at 50 Hz for 10 min (10 cycles of 30s with 1 min interval) at 4°C. The reaction mixture for ATPase consisted of 0.2 ml each of Tris-HClbuffer (0.2 M, pH 7.6), MgCl2 (5 mM), ATP (6 mgmL−1), and sonicated sperm suspension. Different concentrations of SAF (25 μg, 50 μg, and 100 μg) were added to the reaction mixture and incubated at 37°C for 1 h, post which the reaction was stopped by adding 1 ml of cold 10% Trichloroacetic acid (TCA) and then incubated at 4°C overnight for protein precipitation. The control tubes contained all the components of the reaction mixture but TCA was added in the beginning to stop the ATPase activity. Inorganic phosphorus (Pi) released was determined according to the method [11]. One unit of ATPase is expressed as μg of the Pi released after 1 h of incubation.
Effect of intravaginal application of SAF on fertility outcome: in vivo study
To study the effect of SAF on fertility outcome, female BALB/c mice (n=15) synchronised in their oestrous cycles by Whitten effect were divided into three groups (Group I, II and III). Group I was administered intravaginally with single dose of 20 μl PBS and it served as control. Group II and III were intravaginally inoculated with 10 μg and 25 μg of SAF, respectively. For intravaginal inoculation, mice were held on the top of cage in supine position. 20 μl of PBS/SAF was inserted into vaginal lumen with the help of autopipette. The mice were held in same position for a period of 1 min. All the animals were allowed to mate with proven breeder male in ratio 2:1. Next morning, mating was confirmed by presence of vaginal plug. The mice were kept under observation for a period of 35 days and looked for any weight gain, abdominal distension or delivery of pups.
Results
The standard strain of S. marcescens used in the present study was found to cause sperm agglutination in vitro.
Extraction and purification of sperm agglutinating factor (SAF) from Serratia marcescens
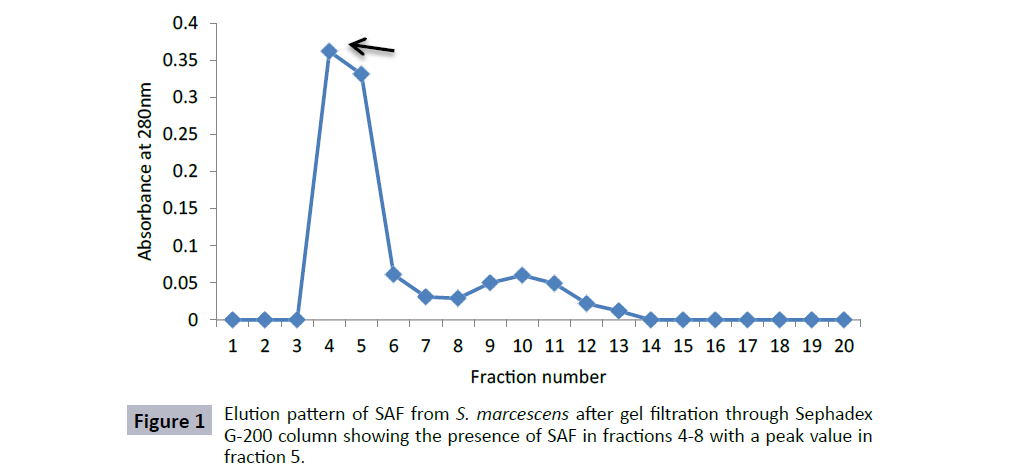
S. marcescens was grown in BHI for 72 h and centrifuged at 10,000 rpm for 10 min at 4°C. The pellet so obtained was dissolved in PBS and sonicated (15 cycle). After sonication, the suspension was centrifuged at 10,000 rpm for 40 min. The supernatant so obtained was collected and was subjected to ammonium sulphate precipitation. The results showed that sperm agglutinating factor could be precipitated out with ammonium sulphate at 60-80% saturation. The ammonium sulphate precipitated, dialysed fraction was subjected to further purification by passing through Sephadex G-200 column. From column chromatographic pattern, it was observed that the agglutinating activity was present in the fractions 4-8 with a peak value in fraction 5, where each fraction was of 3 ml (Figure 1). The fractions showing agglutinating activity were pooled and concentrated using PEG 6000 at 4°C.
DEAE cellulose-ion exchange chromatography
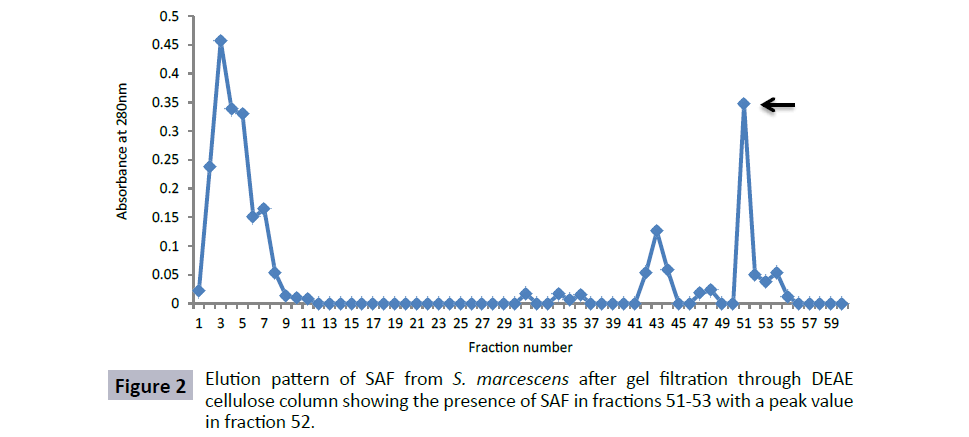
The fractions from Sephadex G-200 column showing agglutination of spermatozoa were pooled, concentrated and applied to DEAEcellulose column. The column was run using step gradient of NaCl with each fraction of 4 ml. It was observed that the protein bound to column could be eluted with PBS containing 0.6 M NaCl. The fractions that showed sperm agglutinating activity were 51- 53 with peak values in fraction 52 (Figure 2). These bioactive fractions were again pooled and concentrated by PEG-6000.
Estimation of molecular weight
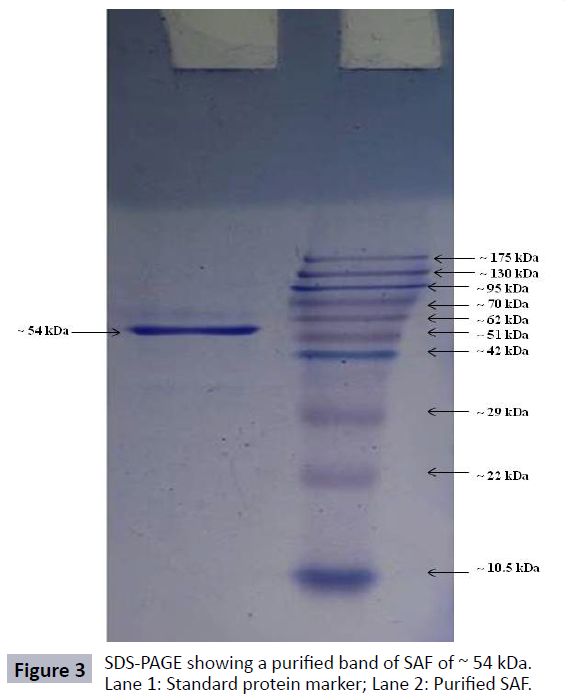
The molecular weight of purified SAF obtained after ion-exchange chromatography was estimated by SDS-PAGE. The results showed the presence of single band indicating that the purified SAF has a molecular weight of approximately 54 kDa (Figure 3).
Impact of SAF on sperm parameters: in vitro studies
Motility
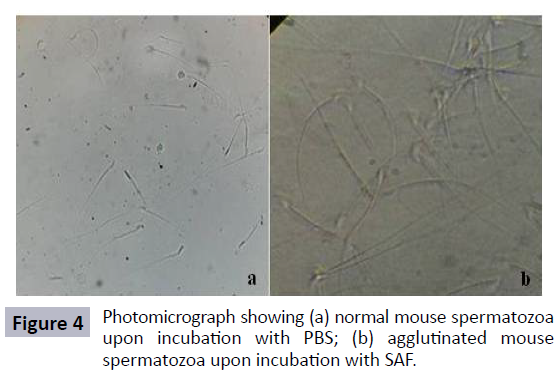
When the effect of SAF was checked on sperm parameters in vitro, it was observed that 60 μg of SAF was able to cause complete agglutination of spermatozoa within 30 min of incubation (Figure 4).
Viability
The effect of SAF on sperm viability was determined by dyeexclusion method using Eosin Y. The results showed that SAF showed spermicidal activity at a concentration of 80 μg within 30 min of incubation (Figure 5).
Mg++ ATPase activity
The effect of SAF on Mg++ ATPase activity of mouse spermatozoa was also evaluated. The results showed that SAF could significantly inhibit Mg++ ATPase activity of spermatozoa since the activity reduced from 1360 units (control) to 700, 320, 150 units in presence of SAF at a concentrations of 25, 50 and 100 μg, respectively (Table 1).
Table 1. Effect of SAF on Mg++ ATPase activity of mouse spermatozoa.
| S. No. | Concentration of SAF (µg) | Units of ATPase (SAF) |
|---|---|---|
| 1 | 25 | 700 |
| 2 | 50 | 320 |
| 3 | 100 | 150 |
Effect of SAF on fertility outcome in female mice: in vivo study
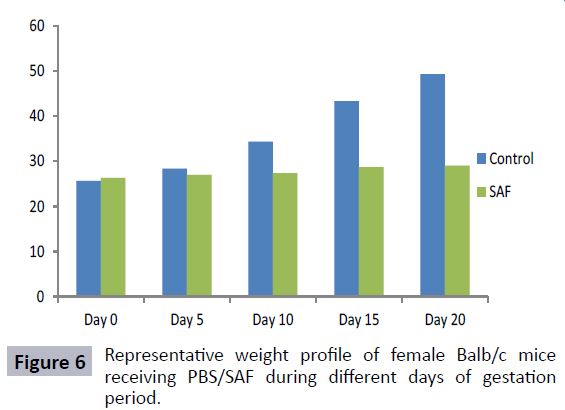
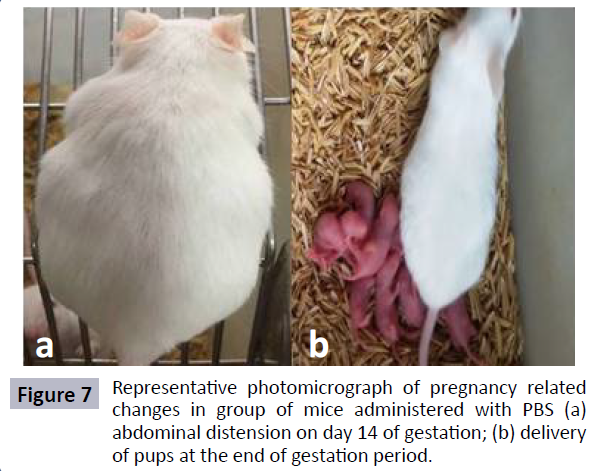
SAF was also evaluated for its effect on fertility outcome of female mice. From the results it could be observed that mice in Group I (receiving 20 μl of PBS) remained fertile as evident by consistent weight gain, abdominal distension and finally delivery of pups at the end of gestation period (Figures 6 and 7). However, in case of mice treated with 10 μg and 25 μg of SAF, conception could be blocked efficiently, as mice did not show any pregnancy related changes and failed to deliver pups at the end of gestation period.
Discussion
Female infertility is a multifaceted health concern which is associated with severe reproductive repercussions [12] It is medically defined as lack of conception after regular unprotected sexual intercourse for a period of one year. Infertility can have negative social and psychological consequences on the individual ranging from more subtle forms of social stigmatization to community banishment or divorce [13]. Different factors that determine the fertility outcome include endocrinological, anatomical and infections of the reproductive tract [14].
Among these potential causes, genitourinary tract infections can be attributed to 30-40% of the cases of infertility [5]. It is widely accepted that infections with sexually transmitted microorganisms viz. Neisseria gonorrheae, Treponemapallidum and Chlamydia trachomatis can lead to fertility alterations [15]. Compared to sexually transmitted microorganisms, little attention has yet been focused on relation between uropathogens and infertility. In the case of common uropathogens, the negative influence on reproduction is suggested but not well established. Abundant in vitro data suggests the detrimental effects of uropathogens on sperm parameters but there is paucity of in vivo relevance of these results [16].
Taking available data into consideration, earlier in our laboratory, blockage of conception in female mice as a result of sperm impairing uropathogens viz. S. aureus, E. coli, P. aeruginosa and S. marcescens has been observed [7,17,18]. Further, the sperm impairing factors isolated and purified from S. aureus and E. coli have also been found to induce infertility. On similar grounds, the present work was carried out with an aim to isolate and purify the sperm agglutinating factor from S. marcescens and study its impact on fertility outcome.
Agglutination of spermatozoa by S. marcescens, was attributed to tight adhesions between bacteria and spermatozoa as the sperm agglutination was the property of washed cells, hence, cell pellet was sonicated in order to extract the spermagglutinating factor (SAF). Purification was done using two step chromatographic techniques using sephadex G-200 and DEAE cellulose.
Earlier, various research groups have isolated and purified sperm impairing factors from different microorganisms. Sperm immobilization factors from E. coli [19,20] S. aureus [21] and extracellular polymeric substance (EPS) from T. vaginalis [22]. Further, a sperm agglutinating factor from S. aureus has also been successfully isolated and purified which is known to cause deterioration of seminal parameters [23].
Sperm motility and viability are considered to be very crucial to the process of fertilization. Any loss in motility and viability of spermatozoa disables their potential to fertilize naturally since they are unable to travel to bind oocyte [24]. In this regard, it was thought to investigate the in vitro effect of SAF on sperm parameters. The results indicate that SAF was able to negatively affect the motility of mouse spermatozoa and rendered the spermatozoa immotile by agglutinating them. Further, SAF not only affected motility of spermatozoa rather it was spermicidal as well. An observation [25] pointed out that release of an unidentified soluble factor by E. coli inhibits mitochondrial membrane potential (ΔΨm), motility and viability of spermatozoa. Similarly, Barbonetti et al. [26] demonstrated that quorum sensing molecules (farnesol and 3-oxododecanoyl-L-homoserine lactone, secreted by Candida albicans and Pseudomonas aeruginosa, respectively), elicit numerous deteriorating effects on spermatozoa.
Spermatozoa are armed with various mechanisms which permit them to reach the female genital tract and fertilize the oocyte. Post reaching the female reproductive tract, the spermatozoa senses the environment and acclimatizes its motility, which is controlled by ATP, whose breakdown is accomplished by cation dependant ATPases to release energy for contractile processes of flagella [27]. To determine the plausibility of involvement of such ATPases in SAF induced sperm impairment, the effect of SAF on Mg++ dependent ATPase was studied. The results, established a negative correlation between the SAF and Mg++dependent ATPase activity suggesting that SAF could possibly have targeted ATPases for imposing an adverse effect on sperm motility. Similar results have been reported by Peralta-Arias et al. [28] in which inhibition of the dynein-ATPase activity led to sperm immobilization. Also, in a recent study, The authors [29,30] attributed the decreased sperm motility in individuals exposed to Cadmium (smokers) to inhibition exerted by Cd2+ on sperm ATPases.
To explore the effect of SAF in vivo, fertility studies were carried out in female Balb/C mice and SAF was administered intravaginally before mating. From the results it was observed that SAF at concentration of ≥ 10 μg led to blockage of conception. It could be interpreted that presence of sperm impairing factors in vagina might alter the female genital tract into a hostile milieu for spermatozoa, which may prevent natural conception and could play a critical role in induction of infertility. Nakano et al. [31] have summarized various parameters such as poor buffering capacity of acid vaginal pH, hormonal dysfunctions and exogenous factors which can interfere with spermatozoal movement through the vagina, therefore, be implicated in the pathophysiology of unexplained infertility.
In conclusion, SAF isolated and purified from S. marcescens was capable of compromising various sperm parameters both in vitro (motility, viability and Mg++ dependent ATPases) and in vivo. Henceforth this sperm agglutinating factor holds enormous antifertility potential and can be exploited as an excellent vaginal contraceptive in future.
References
- Akhi MT, Esmailkhani A, Sadeghi J, Niknafs B, Farzadi L, et al., (2017) The frequency of Staphylococcus aureus isolated from endocervix of infertile women in Northwest Iran. Int J Fertil Steril 11: 28-32.
- Ghiasi M, Fazaeli H, Kalhor N, Sheykh-Hasan M, Tabatabaei-Qomi R (2014) Assessing the prevalence of bacterial vaginosis among infertile women of Qom city. Iran J Microbiol 6: 404-408.
- Inhorn MC, Patrizio P (2015) Infertility around the globe: new thinking on gender, reproductive technologies and global movements in 21st century. Hum Reprod 21: 411-426.
- Souho T, Benlemlih M, Bennani B (2015) Human Papilloma virus infection and fertility alteration: A systematic review. PLoS one 10: e0126936.
- Coskun E, Ozkan S, Vural B (2005) Impact of genital infections on fertility. J Turk Ger Gynecol Assoc 6: 197-203.
- Schuppe HC, Pilatz A, Diemer HHT, Wagenlehner F, Weidner W (2017) Urogenital Infection as a Risk Factor for Male Infertility. Dtsch Arztebl Int 114(19): 339-346.
- Vander H, Prabha V (2015) Evaluation of fertility outcome as a consequence of intravaginal inoculation with sperm-impairing micro-organisms in a mouse model. J Med Microbiol 64: 344-347.
- Emmens CW (1947) The motility and viability of rabbit spermatozoa at different hydrogen ion concentrations. J Physiol 471-481.
- Kielley MW (1955) Mitochondrial ATPase in Methods in Enzymology, Colowick SP, Kaplan NO, editors., pp: 593-595, Academic Press, New York, NY, USA.
- Chappel J B (1963) The effect of alkylguanidines on mitochondrial metabolism. J Biol Chem 238: 410-417.
- Boyce A, Casey A, Walsh G (2004) A phytase enzyme-based biochemistry practical particularly suited to students undertaking courses in biotechnology and environmental science. Biochem Mol Biol Educ32(5): 336-340.
- Campisciano G, Florian F, D’Eustacchio A, Stankovic D, Ricci G, et al., (2017) Subclinical alteration of the cervical-vaginal microbiome in women with idiopathic infertility. J Cell Physiol 232: 1681-1688.
- Cousineau TM, Domar AD (2007) Psychological impact of infertility. Best Pract Res Clin Obstet Gynaecol 21(2): 293-308.
- Kokkayil P, Rawre J, Malhotra N, Dhawan B (2013) Co-infection of Mycoplasma genitalium and Chlamydia trachomatis in an infertile female patient with genital tuberculosis. Indian J Pathol Microbiol 56: 457-459.
- Souho T, Benlemlih M, Bennani B (2015) Human Papilloma virus infection and fertility Alteration: A Systematic Review. PLoS one 10: e0126936.
- Fraczek M, Kurpisz M (2015) Mechanisms of the harmful effects of bacterial semen infection on ejaculated human spermatozoa: potential inflammatory markers in semen. Folia Histochemica Cytobiologica 53:201-217.
- Kaur K, Prabha V (2014) Sperm agglutinating Escherichia coli and its role in infertility: in vivo study. Microb Pathog 70: 33-38.
- Kaur S, Prabha V (2012) Infertility as a consequence of sperm agglutinating Staphylococcus aureus colonization in genital tract of female mice. PLoS one 7: e52325.
- Paulson JD, Polakoski KL (1977) Isolation of a spermatozoal immobilization factor from Escherichia coli filtrates. Fertil Steril 28: 182-185.
- Prabha V, Sandhu R, Kaur S, Kaur K, Sarwal A, et al., (2010) Mechanism of sperm immobilization by Escherichia coli. Adv Urol Vol: 6.
- Prabha V, Gupta T, Kaur S, Kaur N, Kala S, et al., (2009) Isolation of a spermatozoal immobilization factor from Staphylococcus aureus filtrates. Can J Microbiol 55: 874-878.
- Roh J, Lim YS, Seo MY, Choi Y, Ryu JS (2015) The secretory products of Trichomonas vaginalis decrease fertilizing capacity of mice sperm in vitro. Asian J Androl 17: 319-323.
- Ohri M, Prabha V (2005) Isolation of sperm agglutinating factor from Staphylococcus aureus isolated from a woman with unexplained infertility. Fertil Steril 84: 1539-1541.
- Kumar A, Singh A, Ekavali (2015) A review on Alzheimer's disease pathophysiology and its management. Pharmacological Reports 67: 195-203.
- Barbonetti A, Vassallo MRC, Cinque B, Filipponi S, Mastromarino P, et al., (2013) Soluble products of Escherchia coli induce mitochondrial dysfunction-related sperm membrane lipid peroxidation which is prevented by Lactobacilli. PLoS one 8: e83136.
- Rennemeier C, Frambach D, Hennicke F, Dietl J, Staib P (2009) Microbial quorum-sensing molecules induce acrosome loss and cell death in human spermatozoa. Infect Immun 77: 4990-4997.
- Lishko PV (2016) Contraception: Search for an Ideal Unisex Mechanism by Targeting Ion Channel 41: 816-818.
- Peralta-Arias RD, Vivenes CY, Camejo MI, Piñero S, Proverbio T, et al., (2015) ATPases, ion excahnagers and human sperm motility. Reproduction 149(5): 475-484.
- Da Costa R, Botana D, Piñero S, Proverbio F, Marín R (2016) Cadmium inhibits motility, activities of plasma membrane Ca2+-ATPase and axonemal dynein-ATPase of human spermatozoa. Andrologia 48: 464-469.
- Roh J, Lim YS, Seo MY, Choi Y, Ryu JS (2015) The secretory products of Trichomonas vaginalis decrease fertilizing capacity of mice sperm in vitro. Asian J Androl 17: 319-323.
- Nakano FY, Leao RBF, Esteves SC (2015) Insights into the role of cervical mucus and vaginal pH in unexplained infertility. Medical Express 2: 1-8.
Open Access Journals
- Aquaculture & Veterinary Science
- Chemistry & Chemical Sciences
- Clinical Sciences
- Engineering
- General Science
- Genetics & Molecular Biology
- Health Care & Nursing
- Immunology & Microbiology
- Materials Science
- Mathematics & Physics
- Medical Sciences
- Neurology & Psychiatry
- Oncology & Cancer Science
- Pharmaceutical Sciences